

Title image by Charles Ray, Auburn University
This guide to the psocids of Texas is an ongoing project to aid those interested in this often overlooked group. Many specimens, unfortunately, cannot be identified without microscopic examination, dissection and preparation of permanent slides. I present these images to convey the diversity of these remarkable insects. Click the psocid image for a link to that BugGuide species page. Unless otherwise stated, all species' images were taken by the author. Psocids (bark lice and booklice) are now placed with the true lice in the order Psocodea. Psocoptera is no longer a ligitimate taxon name. For convenience, one can put quotes around it and use the term "Psocoptera" to refer to free-living Psocodea.

My "psocid book" is now for sale on my Etsy shop. (Go to etsy.com and search for psocoptera. ) The book is $50 plus shipping. It is my fond hope that the book will appeal to both amateur and professional entomologists. Color images by myself and by participants on iNaturalist and BugGuide make these insects come to life. Scanning electron micrographs reveal the details of psocid anatomy. Psocid reproductive behavior is documented by videos easily accessed by keywords on FigShare.
Title: Adventures in a Minor Order: A citizen scientist explores the biodiversity and behavior of "Psocoptera"- the booklice and barklice.
ISBN: 978-0-9994456-3-1
Publisher: Loflin & Associates, Austin, Texas

The expert on psocids of North America is Dr. Edward L. Mockford of Illinois State University. His book, "North American Psocoptera" (Sandhill Crane Press, 1993), is an essential reference for this group. Imagine the task of taking on an entire order of insects for your life's work! He has described over 430 species! He is a true Renaissance man - fluent in several languages including cladistics. I visited his laboratory where there were boxes of specimens from all over the world. The information listed on this website is taken primarily from Mockford's publications with additional comments from my own personal observations. He is no longer active in psocidology research, but his contribution to our knowledge of "Psocoptera" is beyond measure.

Dr. Alfonso Garcia Aldrete, at the Universidad Nacional Autonoma de Mexico (UNAM), Instituto de Biologia, was a PhD student of Dr. Mockford's and was active in the field of psocidology. He has described over 418 species native to the U.S., Mexico, Central and South America. Many of the tropical psocids are actually colorful and have very unusual wing venation. Dr. Garcia Aldrete certainly had his work cut out for him with hundreds of undescribed species to deal with. His untimely death in 2022 was a great loss to the small group of entomologists who consider themselves psocidologists.




 My most recent psocid adventure began as I opened a box from Ben Gruver of Pawnee County, Oklahoma. There were at least a dozen live Neolepolepis occidentalis in the package. I was interested in documenting courtship and mating behavior in this species. Most North American species in the family Lepidopsocidae are parthenogenetic, so this was my only chance. I was able to successfully rear them and isolate nymphs to provide virgin males and females, but after weeks of effort, I did not succeed. It is difficult to tell males from females in this species, but that was just the beginning of the issues. After more than 50 pairings, I could not break the code.
My most recent psocid adventure began as I opened a box from Ben Gruver of Pawnee County, Oklahoma. There were at least a dozen live Neolepolepis occidentalis in the package. I was interested in documenting courtship and mating behavior in this species. Most North American species in the family Lepidopsocidae are parthenogenetic, so this was my only chance. I was able to successfully rear them and isolate nymphs to provide virgin males and females, but after weeks of effort, I did not succeed. It is difficult to tell males from females in this species, but that was just the beginning of the issues. After more than 50 pairings, I could not break the code.
Meanwhile, Ben was also rearing them and documenting behavior. He has photographed eggs of this species and they look very much like those of Echmepteryx hageni. In one of his rearing containers, a nymph appeared with longer than usual wing pads. Ben had reared the rare macropterous form of this species. It's quite a beauty as you can see from Ben's image. Perhaps he will also encounter a macropterous male!
 My barklice adventures have just begun. As you can see, I get much help from my feline companion. My range maps, which are actually record maps, are taken from my own collection, from specimens recorded from the University of Texas Insect Collection, Texas A&M Insect Collection, from the scientific literature and even BugGuide and iNaturalist. As I extend the ranges, I will update the range maps. I will be adding species to the website as I encounter them. Please
inform me if you have preserved specimens with collection data that would add counties to the Texas range maps. Range maps not shown for Liposcelididae. Species identification in this taxon is very difficult and I will not attempt to post images unless I have had expert verification. Undescribed species may not have a BugGuide page. Dissection of psocids and preparation of permanent slides is a character-building experience for sure. With practice, however, one does do what first seemed impossible.
My barklice adventures have just begun. As you can see, I get much help from my feline companion. My range maps, which are actually record maps, are taken from my own collection, from specimens recorded from the University of Texas Insect Collection, Texas A&M Insect Collection, from the scientific literature and even BugGuide and iNaturalist. As I extend the ranges, I will update the range maps. I will be adding species to the website as I encounter them. Please
inform me if you have preserved specimens with collection data that would add counties to the Texas range maps. Range maps not shown for Liposcelididae. Species identification in this taxon is very difficult and I will not attempt to post images unless I have had expert verification. Undescribed species may not have a BugGuide page. Dissection of psocids and preparation of permanent slides is a character-building experience for sure. With practice, however, one does do what first seemed impossible.
I consider myself a citizen scientist now that I'm retired. I was an entomology major for my first 3 years of college but then, due to circumstances too numerous to mention, spent my career in biochemistry. My passion was always bugs and now I have returned to that noble quest. Macro photography is essential when working with tiny barklice. To the left is my "horizontal" setup and to the right my "vertical" one. I post regularly on BugGuide and find it not only an incomparable source of information but a link to others who share my love of insects.
When one is out collecting with a beat sheet people as well as dogs are perplexed. What is that thing they ask? The next question, from people, is: Why? or the dreaded "What are they good for? What a thrill to find something totally "new". One can do that with amateur entomology.
Psocids include barklice, found in trees, shrubs and leaf litter as well as booklice which inhabit man-made dwellings and dead vegetation. They are hemimetabolous (incomplete metamorphosis) and share with other hemipteroid insects a reduction in tarsomeres, absence of cerci, and unique anatomy of the hypopharynx. These insects were until recently placed in the order Psocoptera with the lice in a separate order Phthiraptera. Molecular genetic evidence has confirmed past suspicions that true lice evolved from booklice and this finding has brought about a taxonomic reshuffling in which bark lice and parasitic lice share the order Psocodea. According to K. Yoshizawa, C. Lienhard and K. Johnson, "seven suborders are now generally recognized within the Psocodea: Trogiomorpha (bark lice), Troctomorpha (book lice and bark lice), Psocomorpha (bark lice), Amblycera (chewing lice), Ischnocera (chewing lice), Rhynchophthirina (chewing lice) and Anopleura (sucking lice)"; Zool. J. Linn. Soc. 146:287-299, 2006.
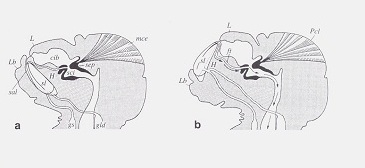 Lingual sclerites Soft bodied psocids and true lice are surprisingly resistant to desiccation because of a remarkable ability described by D. Rudolph (J. Insect Physiol. 28(3):205-212, 1982). As shown in this illustration from Rudolph's paper, psocids can eject "lingual sclerites" to the external environment where they actively absorb water from humid air. These structures are then pulled back into the head. This ability is also present in booklice and true lice and helps explain their presence in relatively arid habitats.
Lingual sclerites Soft bodied psocids and true lice are surprisingly resistant to desiccation because of a remarkable ability described by D. Rudolph (J. Insect Physiol. 28(3):205-212, 1982). As shown in this illustration from Rudolph's paper, psocids can eject "lingual sclerites" to the external environment where they actively absorb water from humid air. These structures are then pulled back into the head. This ability is also present in booklice and true lice and helps explain their presence in relatively arid habitats.
I had never seen the lingual sclerites until videotaping mating behavior in Amphigerontia montivaga. After the male and female separated the female began manipulating the spermatophore to extract the spermatozoa inside. This effort must have been exhausting. She extruded a bright orange lingual sclertite. Wonder what chemical process allows this structure to absorb water so efficiently?
To view see Video # 1 (at 2:50 min.) at the end of the Anatomy section.
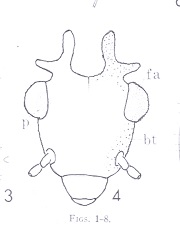
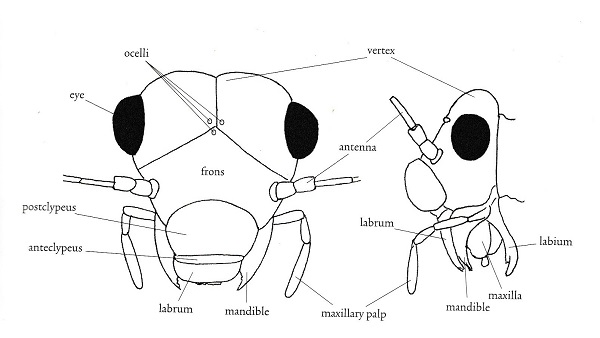 Morphological terms in entomology vary with the taxon under consideration. I'm including this section as an aid to those brave enough to attempt to key out psocid specimens. It is also my hope that you will find some of the items here interesting. First I have included two figures from The Psocoptera of Tropical South-east Asia by T. R. New & C. Lienhard - depicting the anatomy of the head and wings. For more detail see the chapter on Psocoptera by E. L. Mockford in Borror and DeLong's Introduction to the Study of Insects by Triplehorn and Johnson. All scanning electron microscopy (SEM) images were taken by Gregory Paulson of Shippensburg University.
Morphological terms in entomology vary with the taxon under consideration. I'm including this section as an aid to those brave enough to attempt to key out psocid specimens. It is also my hope that you will find some of the items here interesting. First I have included two figures from The Psocoptera of Tropical South-east Asia by T. R. New & C. Lienhard - depicting the anatomy of the head and wings. For more detail see the chapter on Psocoptera by E. L. Mockford in Borror and DeLong's Introduction to the Study of Insects by Triplehorn and Johnson. All scanning electron microscopy (SEM) images were taken by Gregory Paulson of Shippensburg University.
"The evolutionary history of the Paraneoptera is beautifully reflected in structure and function of their mouthparts. There is a general trend from the most generalized 'picking' mouthparts of Psocoptera with standard insect mandibles, to the probing and puncturing mouthparts of thrips and anapleuran lice, and the distinctive piercing-sucking rostrum or beak of the Hemiptera." Evolution of Insects, D. Grimaldi & M. Engel, 2005, p. 261. In psocids, the labial palps are redued to 2 segments in Trigiomorpha and Troctomorpha and a single one in Psocomorpha.

 Lacinia The lacina of the maxilla in psocids have evolved into an elongate pick-like structures unique to the "Psocopera". Lacinia are thought to
scrape out food material but are also thought to prop the head while the mandibles are in use.
Lacinia The lacina of the maxilla in psocids have evolved into an elongate pick-like structures unique to the "Psocopera". Lacinia are thought to
scrape out food material but are also thought to prop the head while the mandibles are in use.
These structures are beautifully depicted on the SEM image to the left from Amphigerontia montivaga. Going from anterior to posterior are the labrum, mandibles, lacinia, and maxillary palps; then the labium with the opening of the silk gland along the midline. That is the long backwards-pointing structure. For a close up view see section on silk. To the right is an image from Tapinella maculata with a clearer view of the mandibles and laciniae. The laciniae look like toothpicks with prongs.
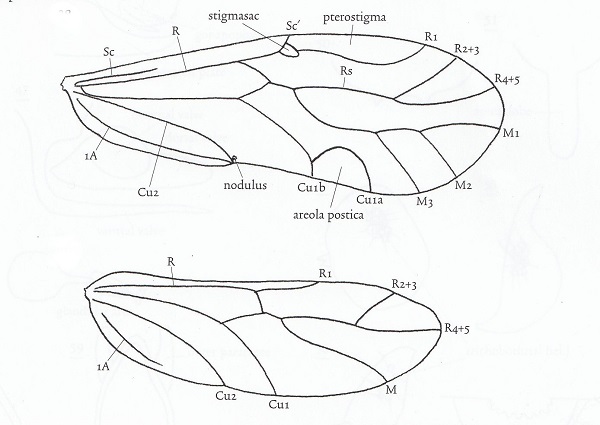 Wing venation and other anatomical features vary considerably among families. Individuals may be macropterous, brachypterous, micropterous or apterous, depending on species and environmental or phenological conditions. Apterous and micropterous psocids lack ocelli. Alary polymorphism occurs in many species. In the family Pachytroctidae, for example, males are apterous and females either apterous or macropterous. Sexual dimorphism in Mesopsocus results in macropterous males and apterous females. The dimorphism is reversed in Embidopsocus.
Wing venation and other anatomical features vary considerably among families. Individuals may be macropterous, brachypterous, micropterous or apterous, depending on species and environmental or phenological conditions. Apterous and micropterous psocids lack ocelli. Alary polymorphism occurs in many species. In the family Pachytroctidae, for example, males are apterous and females either apterous or macropterous. Sexual dimorphism in Mesopsocus results in macropterous males and apterous females. The dimorphism is reversed in Embidopsocus.
Don't get the impression that the wing diagram above will work for all psocids. Far from it. When the aerola postica fuses with vein M, as in the family Psocidae, the cell created is called the discoidal cell.The wings of members of the family Lepidopsocidae are covered with scales which look much like those of lepidopterans. Wing venation varies widely in different familes of Psocoptera from minimal in Archipsocidae and Liposcelididae to very complex in the tropical family Calopsocidae. Supernumerary veins, for example, result in a very complex pattern in Ianthorntonia annae (Epipsocidae) of Brazil which is named after the noted psocidologist Ian Thornton.
Psocids are unusual among insects in that they have two different wing-coupling mechanisms. One anchors the wings at rest and the other couples the wings in flight.
 Nodus The stigmapophysis (also known as the stigmasac or nodus) at the base of the pterostigma engages the costal vein of the hind wing and locks the wings into a "tent" when at rest in members of Psocomorpha. In the more basal Trigiomorpha and Troctomorpha, the wings are held flat. According to Ogawa and Yoshizawa (Arthropod Structure & Development, 47(4): 416-422, 2018) this structure apparently evolved twice within the "Psocoptera".
Nodus The stigmapophysis (also known as the stigmasac or nodus) at the base of the pterostigma engages the costal vein of the hind wing and locks the wings into a "tent" when at rest in members of Psocomorpha. In the more basal Trigiomorpha and Troctomorpha, the wings are held flat. According to Ogawa and Yoshizawa (Arthropod Structure & Development, 47(4): 416-422, 2018) this structure apparently evolved twice within the "Psocoptera".

 Nodulus refers to the region on the posterior margin of the hindwing where veins Cu2 and 1A join. This is where the in-flight wing-coupling mechanism is located. The coupling mechanism arose in the common ancestor of Psocodea and is present in all three suborders of "Psocoptera" but is secondarily lost in Nanopsocetae (Liposcelididae and Pachytroctidae) Two SEM images depict this wing-coupling structurre. Look carefully at the image to the left and you will see the hook-like retinaculum engaging the costal vein of the hind wing. To the right one can see the retinaculum and the enlarged adjacent hind margin, the retainer. These two structures act in concert to grasp the costal vein of the hind wing. N. Ogawa and K. Yoshizawa recently published a comprehensive study of the evolution of this mechanism which is unique to "Psocoptera" (Origin and transformation of the in-flight wing-coupling structure in Psocodea. J. of Morphology 279(4):517-530, 2018). In my humble opinion, this SEM image (right) of the nodulus is a work of art! I think I'm going to frame it.
Nodulus refers to the region on the posterior margin of the hindwing where veins Cu2 and 1A join. This is where the in-flight wing-coupling mechanism is located. The coupling mechanism arose in the common ancestor of Psocodea and is present in all three suborders of "Psocoptera" but is secondarily lost in Nanopsocetae (Liposcelididae and Pachytroctidae) Two SEM images depict this wing-coupling structurre. Look carefully at the image to the left and you will see the hook-like retinaculum engaging the costal vein of the hind wing. To the right one can see the retinaculum and the enlarged adjacent hind margin, the retainer. These two structures act in concert to grasp the costal vein of the hind wing. N. Ogawa and K. Yoshizawa recently published a comprehensive study of the evolution of this mechanism which is unique to "Psocoptera" (Origin and transformation of the in-flight wing-coupling structure in Psocodea. J. of Morphology 279(4):517-530, 2018). In my humble opinion, this SEM image (right) of the nodulus is a work of art! I think I'm going to frame it.
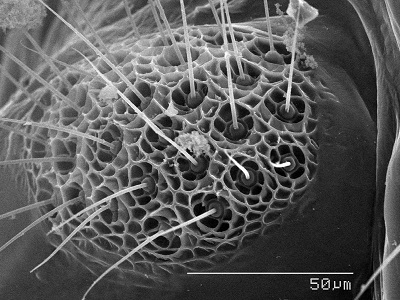 Sensorium One of the most fascinating anatomical features of barklice is the
sensorium or sense cushion which is found on the dorso-lateral surface of the paraproct in winged specimens. This structure is thought to be a reminant of the
cerci. It consists of closely set sense organs, each consisting of a sensory "hair" surrounded by a rosette or "basal floret". One can imagine the hair being moved by a gust of wind and triggering a direction-specific nerve ending as the basal floret is warped. The SEM image to the right depicts the sensorium with
trichobothria surrounded by basal florets. It is theorized that this structure detects wind currents and aids in flight. Each sensory unit is called a trichobothrium and the number of trichobothria is species specific. In more "primitive" species the sensorium has fewer more dispersed trichobothria. Speleketor flocki (Prionoglaridae), for example, has only 4 trichobothria/ sensorium. Ref.: Mockford, E. L., The Southwestern
Naturalist 29(2): 169-179, 1984.
Sensorium One of the most fascinating anatomical features of barklice is the
sensorium or sense cushion which is found on the dorso-lateral surface of the paraproct in winged specimens. This structure is thought to be a reminant of the
cerci. It consists of closely set sense organs, each consisting of a sensory "hair" surrounded by a rosette or "basal floret". One can imagine the hair being moved by a gust of wind and triggering a direction-specific nerve ending as the basal floret is warped. The SEM image to the right depicts the sensorium with
trichobothria surrounded by basal florets. It is theorized that this structure detects wind currents and aids in flight. Each sensory unit is called a trichobothrium and the number of trichobothria is species specific. In more "primitive" species the sensorium has fewer more dispersed trichobothria. Speleketor flocki (Prionoglaridae), for example, has only 4 trichobothria/ sensorium. Ref.: Mockford, E. L., The Southwestern
Naturalist 29(2): 169-179, 1984.
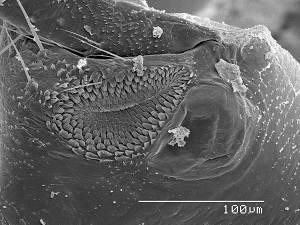 Rasp & mirror The hind coxae of many adult psocids bears a rasp and mirror otherwise known as Pearman’s organ. The rasp is a denticulate field and the mirror a minute tympanum. It is thought that the rasps of opposing coxae are rubbed together to produce a sound which is amplified by the adjacent mirrors. It is more complex in winged members of the Psocomorpha and is better developed in males. The SEM image to the left is the rasp and mirror of Hyalopsocus striatus. What a shame that so little research has been done to verify these ideas. It’s just another case of an overlooked and under-appreciated taxon. In the following video (see link below) you will see possible stridulation behavior on the part of the male and female of Amphigerontia montivaga. The male moves very fast, so his behavior is more difficult to interpret. It sure looks to me as though the female could be rubbing her hind coxae togeather. In this video notice that the spermatophore passes from the male to female. Also notice the bright orange lingual sclerite which protrudes from the mouth of the female as she manipulates the spermatophore to recover the sperm inside. View full screen for best results.
Rasp & mirror The hind coxae of many adult psocids bears a rasp and mirror otherwise known as Pearman’s organ. The rasp is a denticulate field and the mirror a minute tympanum. It is thought that the rasps of opposing coxae are rubbed together to produce a sound which is amplified by the adjacent mirrors. It is more complex in winged members of the Psocomorpha and is better developed in males. The SEM image to the left is the rasp and mirror of Hyalopsocus striatus. What a shame that so little research has been done to verify these ideas. It’s just another case of an overlooked and under-appreciated taxon. In the following video (see link below) you will see possible stridulation behavior on the part of the male and female of Amphigerontia montivaga. The male moves very fast, so his behavior is more difficult to interpret. It sure looks to me as though the female could be rubbing her hind coxae togeather. In this video notice that the spermatophore passes from the male to female. Also notice the bright orange lingual sclerite which protrudes from the mouth of the female as she manipulates the spermatophore to recover the sperm inside. View full screen for best results.
Video # 1 Watch on youtube
 Among the few entomologists who have dared venture into "Psocoptera", there have been some real characters. Although known as an expert on dragonflies, Robin Tillyard described numerous psocid species, including some ancient ones. He was obsessed by the giant Permian griffenflies. According to David Grimaldi and Michael Engel (Evolution of the Insects, p.140) "Tillyard sought a mystic to reveal the deep past so that he could see the griffenflies in flight, but he was a careful and thoughtful scientist." Legend has it that Tillyard gave a lecture in the 1920s while visiting Kansas and members of the audience said that he spoke of the griffenflies as though he had seen them in person. The lower Permian Elmo limestone formation of Kansas has yielded fossil dragonflies as well as insects which were "well on their way to being Psocoptera" (Courtney N. Smithers, Classification and Phylogeny of the Psocoptera, Part V. Fossil Psocoptera, The Australian Museum Memoir 14, 1972, p. 240). Image to the right shows Tillyard (center) examining fossils from Elmo (courtesy of Roy J. Beckemeyer, windsofkansas.com). This is one of several newly discovered images of Tillyard.
Among the few entomologists who have dared venture into "Psocoptera", there have been some real characters. Although known as an expert on dragonflies, Robin Tillyard described numerous psocid species, including some ancient ones. He was obsessed by the giant Permian griffenflies. According to David Grimaldi and Michael Engel (Evolution of the Insects, p.140) "Tillyard sought a mystic to reveal the deep past so that he could see the griffenflies in flight, but he was a careful and thoughtful scientist." Legend has it that Tillyard gave a lecture in the 1920s while visiting Kansas and members of the audience said that he spoke of the griffenflies as though he had seen them in person. The lower Permian Elmo limestone formation of Kansas has yielded fossil dragonflies as well as insects which were "well on their way to being Psocoptera" (Courtney N. Smithers, Classification and Phylogeny of the Psocoptera, Part V. Fossil Psocoptera, The Australian Museum Memoir 14, 1972, p. 240). Image to the right shows Tillyard (center) examining fossils from Elmo (courtesy of Roy J. Beckemeyer, windsofkansas.com). This is one of several newly discovered images of Tillyard.
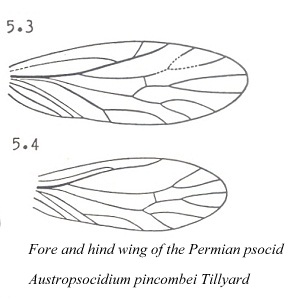 The ancient "psocids" described by Tillyard from Permian formations in Kansas and Australia differed from modern psocids in the following characters: 1) fore and hind wings were of more equal size, 2) wing venation was more extensive; for example, vein M had 4 branches, 3) antennae had more than 50 segments, 4) tarsi were 4-segmented, and 5) maxillary and labial palps had greater number of segments.
The ancient "psocids" described by Tillyard from Permian formations in Kansas and Australia differed from modern psocids in the following characters: 1) fore and hind wings were of more equal size, 2) wing venation was more extensive; for example, vein M had 4 branches, 3) antennae had more than 50 segments, 4) tarsi were 4-segmented, and 5) maxillary and labial palps had greater number of segments.
According to Charles Lienhard and Courtney N. Smithers in "Psocoptera World Catalogue & Bibliography", Instrumenta Biodiversitatis , Museum d'hostoire naturelle, Geneve, 2002 : The [Permian] fossil "psocids" cannot be assigned to Psocoptera as this order is defined at present and are not mentioned in this catalogue." Cenozoic and Cretaceous psocid species found in amber are included in this catalogue.
 "Psocoptera" are well represented as inclusions in amber. Baltic amber was formed during the Eocene epoch at around 44 million years ago. The image to the left (Baltic) is from a beautiful website by Anders Damgaard.
"Psocoptera" are well represented as inclusions in amber. Baltic amber was formed during the Eocene epoch at around 44 million years ago. The image to the left (Baltic) is from a beautiful website by Anders Damgaard.
David Grimaldi and Michael Engel (Fossil Liposcelididae and the lice ages, Proc. R. Soc. B 273:625-633, 2005) describe the "oldest and most primitive Liposcelididae" from mid-cretaceous amber (100 million year old) from Myanmar. Cretoscelis burmitica possessed numerous plesiomorphic traits that distinguish it from all other genera of winged Liposcelididae. This find extends the geological age of the family to twice that of the previous oldest fossil Embidopsocus eocenicus. The authors speculate on the origin of lice: "we find louse polyphyly implausible for several reasons. ...[it] requires two origins of a suite of features distinctive to lice. "Until compelling evidence indicates otherwise, it seems reasonable to assume monophyly of Phthiraptera. This specialized lifestyle apparently originated in the Early Cretaceous, fed by diverse early mammals, haired pterosaurs, and feathered thropods. Phthiraptera probably did not significantly diversify, though, until the large radiations of placental mammals and passerine birds that took place in the Tertiary, which can justifiably be called an 'age of lice'".
Tim King of Highland Lake (Blount Co, Alabama) recently discovered psocids using a Berlese funnel on leaf litter which were the first living species of a genus previously only known from Baltic amber. In a sense they are "living fossils". Edward Mockford recently described the species Sphaeropsocus bicolor, A new species of Sphaeripsocus Hagen from southeastern United States: the first living species of its genus, Life: The Excitement of Biology,1(2):100-111, 2013).
In a recent study of all fossil "Psocoptera" known to occur in amber, 32 species were listed encompassing 27 genera and 11 families, including the extinct Archaetropidae and Empheriidae (Mockford, E. L.; Lienhard, Charles; and Yoshizawa, Kazunori; Revised Classification of "Psocoptera" from Cretaceous Amber, a reassessment of published information", Insecta Matsumurna, New Series 69:1-26, 2013). No extant genus was represented in Cretaceous amber. The Psocomorpha represent 6% of species in Cetaceous amber, 69% in Baltic amber (Paleogene) and 84% in extant species. The main radiation of the Psocomopha, therefore, appears to have occurred in the Cenozoic.
 Silk is only produced in the Classes Insecta and Arachnida. This proteinaceous material is composed of repetitive amino acid sequences and transforms from liquid to solid form as it is extruded. "Despite the fact that silks spun by insects and spiders evolved independently, they display some remarkable similarities that could suggest convergence and stabilizing selection" Catherine L. Craig, "Spiderwebs and Silk", Oxford U. Press, 2003, p.32. Silk production, in at least some species, is present in all insect groups except Collembola, Protura, Odonata, Phthiraptera and Zoraptera. Even larval fleas can spin silk. "On the basis of silk gland type, silk protein molecular structure, and the phylogenetic relationship of silk-producing species, we grouped insect silks into 23 distinct categories, each likely to represent an independent evolutionary event. Despite having diverse functions and fundamentally different protein structures, these silks typically have high levels of protein crystallinity and similar amino acid compositions. The substantial crystalline content confers extraordinary mechanical properties and stability to silk and appears to be required for production of fine protein fibers." T. D. Sutherland, J.H. Young, S. Weisman, C.Y. Hayashi and D. J. Merritt, Insect Silk: One Name, Many Materials, Ann. Rev. Entomol. 55:171-188, 2010.
Silk is only produced in the Classes Insecta and Arachnida. This proteinaceous material is composed of repetitive amino acid sequences and transforms from liquid to solid form as it is extruded. "Despite the fact that silks spun by insects and spiders evolved independently, they display some remarkable similarities that could suggest convergence and stabilizing selection" Catherine L. Craig, "Spiderwebs and Silk", Oxford U. Press, 2003, p.32. Silk production, in at least some species, is present in all insect groups except Collembola, Protura, Odonata, Phthiraptera and Zoraptera. Even larval fleas can spin silk. "On the basis of silk gland type, silk protein molecular structure, and the phylogenetic relationship of silk-producing species, we grouped insect silks into 23 distinct categories, each likely to represent an independent evolutionary event. Despite having diverse functions and fundamentally different protein structures, these silks typically have high levels of protein crystallinity and similar amino acid compositions. The substantial crystalline content confers extraordinary mechanical properties and stability to silk and appears to be required for production of fine protein fibers." T. D. Sutherland, J.H. Young, S. Weisman, C.Y. Hayashi and D. J. Merritt, Insect Silk: One Name, Many Materials, Ann. Rev. Entomol. 55:171-188, 2010.
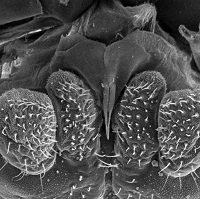 Among insects there are four distinct types of glands that produce silk: collateral, dermal, labial and malpighian. Labial silk glands evolved only twice, once in the "Psocoptera" and later in holometabolos insect larvae (Craig., p.19). The simplified labium in psocids consists of labial palps of only one to two segments with the silk gland opening between them. Psocids often cover their eggs with a sparse layer of silk. I have personally observed lachesillids spin silk in my collection vials and I suspect that some species might use silk in some escape strategy but this is just wild speculation on my part. Both nymphs and adult psocids can spin silk. To the right is a close up of the external opening of the silk gland on the labium. It's the pointed structure along the midline.
Among insects there are four distinct types of glands that produce silk: collateral, dermal, labial and malpighian. Labial silk glands evolved only twice, once in the "Psocoptera" and later in holometabolos insect larvae (Craig., p.19). The simplified labium in psocids consists of labial palps of only one to two segments with the silk gland opening between them. Psocids often cover their eggs with a sparse layer of silk. I have personally observed lachesillids spin silk in my collection vials and I suspect that some species might use silk in some escape strategy but this is just wild speculation on my part. Both nymphs and adult psocids can spin silk. To the right is a close up of the external opening of the silk gland on the labium. It's the pointed structure along the midline.
Video # 2 A female Ectopsocus meridionalis silking over her clutch of eggs: here.
The champion among psocids, as far as silk spinning, occurs in the Archipsocidae, the basal Psocomorph lineage. Archipsocus nomas, for example, builds extensive silken retreats like that shown to the left in a photo by Dr. Douglas Caldwell of the University of Florida. The webbing is harmless but often attracts the attention of homeowners who immediately call the exterminator. What a shame.

 Let’s face it—psocids are not known for their bright colors except when you enter the Tropics. Our North American species pretty much stick to earth tones. Many blend in simply by their coloration as does this psyllipsocid (to the left) against the mortar of a brick wall. This image was taken by John Schneider of Houston. Many cases of such cryptic coloration allow psocids to virtually disappear on bark, lichen, dead leaves or rock outcrops.
Let’s face it—psocids are not known for their bright colors except when you enter the Tropics. Our North American species pretty much stick to earth tones. Many blend in simply by their coloration as does this psyllipsocid (to the left) against the mortar of a brick wall. This image was taken by John Schneider of Houston. Many cases of such cryptic coloration allow psocids to virtually disappear on bark, lichen, dead leaves or rock outcrops.
Many nymphs in the genera Blaste and Loensia (family Psocidae) are covered with blunt-tipped hairs which cause debris to stick and mask their presence. Can you find the Loensia moesta nymph to the right? The remarkable SEM images by Gregory Paulson (below) show the lengths to which these creatures go to provide camouflage. He took these SEM images of a nymph of the same species. One wonders whether these hairs secrete some kind of glue. Notice that even the compound eyes, wings and legs have these sticky hairs. Mockford says in North American Psocoptera, p. 3: "Nymphs of some species of Psocomorpha have gland hairs, short setae with flaring tips. These in some cases retain bits of debris, thus forming a camouflaging coat over the body surface. In other cases, the gland hairs may facilitate body contact among nymphs".

A well documented case of industrial melanism was reported in Mesopsocus unipunctatus (C.Popescu, E. Broadhed and B. Shorrocks. Ecological Entomology 3:209-219, 1978). In this species dark individuals predominated in industrial regions of Yorkshire, while a light form predominated in the Yorkshire Moors and Dales. It would be interesting to follow up on this report and determine whether the dark form has declined with increased pollution regulations.

There are even some species that possibly camouflage by mimesis, or appearing to be something else. Take Camelopsocus monticolus Mockford (on the left) in which the nymphs and wingless females, when perched on a stem, might resemble thorns to potentiel predators. Tree hoppers aren't the only insects with this trick up their sleeves. C. bactrianus, C. hiemalis and C. similis, all North american species, have similar abdominal anatomy and might utilize the same strategy. This is all speculation on my part as Ed Mockford who described these species makes no mention of this apect of thier biology.
Psocids also employ what could be considered disruptive coloration. Myopsocids are the North American champions at cloaking their identity. Even thier compound eyes have pigment patterning that makes them less obvious. This specimen of Lichenomima (to the right) is hard to spot on bark. Even the genus name hints at thier talent for disappearing on a lichen-covered branch.
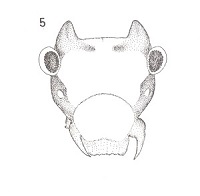
 This little horned devil, Peritroctes bengalensis Thornton & Wong hails from the botanical gardens of Calcutta (Thornton, W. B. & S. K. Wong, Some Psocoptera from West Bengal, India, Transactions of the Royal Entomological Society of London, 118(1): 1-21, 1966). This species is in the family Pachytroctidae. Now here is the amazing thing that happened. Right after I read about this curious creature, I found some living in my leaf liter/ dead palm frond habitat!! I did not know they even occured on this continent. Dr. Mockford was a bit surprised as well and he told me that he and his PhD student Alfonso Gracia Aldrete are currently working on Pachytroctidae. Small world, isn't it? I am sending him specimens of the male and winged female of the little horned devil. Amateur entomology can be so much fun!
This little horned devil, Peritroctes bengalensis Thornton & Wong hails from the botanical gardens of Calcutta (Thornton, W. B. & S. K. Wong, Some Psocoptera from West Bengal, India, Transactions of the Royal Entomological Society of London, 118(1): 1-21, 1966). This species is in the family Pachytroctidae. Now here is the amazing thing that happened. Right after I read about this curious creature, I found some living in my leaf liter/ dead palm frond habitat!! I did not know they even occured on this continent. Dr. Mockford was a bit surprised as well and he told me that he and his PhD student Alfonso Gracia Aldrete are currently working on Pachytroctidae. Small world, isn't it? I am sending him specimens of the male and winged female of the little horned devil. Amateur entomology can be so much fun!
I later found out that there is another bizarre species in the same family that does it one better. Ashley B. Gurney in 1965 described Antilopsocus nadleri from Trinidad and Brazil. "I am glad to name this remarkable insect in honor of one of its discoverers, my friend Aaron M. Nadler" from "A New Genus of Neotropical Psocids with Horn-like structures on the Head", Entomological News, 76(1): 1-10, 1965. One wonders if the male of the species (currently unknown) also has "antlers". Might these decorations on the vertex be part of a mating ritual? Could these amazing "antlers" be an example of sexual selection even if both sexes might bear them? If I'm ever down that way, I'd sure like to see one of these first hand!
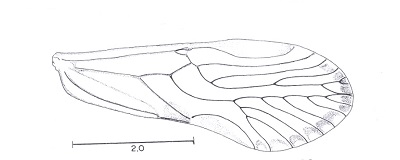 The supernumerary veins in the forewing of members of the new genus Ianthorntonia really are super! This genus is named after the psocidologist Ian Thornton. These Bolivian psocids are in the family Epipsocidae which is one of the most diverse in the "Psocoptera". See Alfonso N. Garcia Aldrete, "A new epiposcid genus from Bolivia", in Thorntonia A Commemorative Volume for Ian W. B. Thornton, Publicaciones Especiales 20, Universiad Nacional Autonoma de Mexico, pp. 99-113, 2004.
The supernumerary veins in the forewing of members of the new genus Ianthorntonia really are super! This genus is named after the psocidologist Ian Thornton. These Bolivian psocids are in the family Epipsocidae which is one of the most diverse in the "Psocoptera". See Alfonso N. Garcia Aldrete, "A new epiposcid genus from Bolivia", in Thorntonia A Commemorative Volume for Ian W. B. Thornton, Publicaciones Especiales 20, Universiad Nacional Autonoma de Mexico, pp. 99-113, 2004.
 Virtually all psocids are oviparous. The only published exceptions are in the genus Archipsocopsis. In Archipsocopsis fernandi, embryos remain in the ovarian tubules until "birth". The eggs contain no yolk and embryos are nurished by the serosal lining of the ovarian tubule Quart. J. Micr. Sci. 77: 99-119, 1934). Mockford also observed viviparity in Archipsocopsis parvulus (Bull. Fla. State Museum 1(5): 253-274, 1957).
Virtually all psocids are oviparous. The only published exceptions are in the genus Archipsocopsis. In Archipsocopsis fernandi, embryos remain in the ovarian tubules until "birth". The eggs contain no yolk and embryos are nurished by the serosal lining of the ovarian tubule Quart. J. Micr. Sci. 77: 99-119, 1934). Mockford also observed viviparity in Archipsocopsis parvulus (Bull. Fla. State Museum 1(5): 253-274, 1957).
Psocid eggs may be laid singly or in groups, the number being dependent on the number of ovarioles in the ovary. Eggs may be encrusted with chewed and only partly digested material as in Tapiella maculata, Ptycta polluta and Trichadenotecnum. In Blaste quieta, a mass of eggs is laid then covered with chewed and partly digested bark, making the eggs difficult to detect. Many species cover their eggs with silk as seen in Valenzuela flavidus and Stenocaecilius casarum. Psocid eggs can enter diapause during winter or periods of extreme heat.
 Contrary to earlier reports, psocid eggs do indeed possess micropyles which allow sperm to enter at fertilization. As in other insects, the egg is oriented in the oviduct in such a way that the micropylar region is in the proximity of the spermathecal duct as it passes through the oviduct. Zuzana Kucerova (Eur. J. Entomol. 99:491-503, 2002), using scanning electron microscopy, found a micropylar region on the eggs of all 13 of the synanthropic species she investigated. These domestic species included Trogium pulsatorium, Lepinotus patruelis, Dorypteryx domestica, Psyllipsocus ramburii, Liposcelis decolor, Liposeclis bostrichophila, and Lachesilla pedicularia. Presented here are previously unpublished SEM images by Gregory Paulson depicting a series of eggs (uper panel) and micropylar regions (lower panel) of several species of Lachesilla. One wonders how the sperm manage to locate this tiny region on the egg.
Contrary to earlier reports, psocid eggs do indeed possess micropyles which allow sperm to enter at fertilization. As in other insects, the egg is oriented in the oviduct in such a way that the micropylar region is in the proximity of the spermathecal duct as it passes through the oviduct. Zuzana Kucerova (Eur. J. Entomol. 99:491-503, 2002), using scanning electron microscopy, found a micropylar region on the eggs of all 13 of the synanthropic species she investigated. These domestic species included Trogium pulsatorium, Lepinotus patruelis, Dorypteryx domestica, Psyllipsocus ramburii, Liposcelis decolor, Liposeclis bostrichophila, and Lachesilla pedicularia. Presented here are previously unpublished SEM images by Gregory Paulson depicting a series of eggs (uper panel) and micropylar regions (lower panel) of several species of Lachesilla. One wonders how the sperm manage to locate this tiny region on the egg.
 Psocid eggs do play host to parasitoid wasps of the family Mymaridae, which lay their eggs in recently deposited host eggs. One remarkable such "fairyfly" was described by E. L. Mockford (Ann. Entomol. Soc. Am. 90(2): 115-120, 1997). Dicopomorpha echmepterygis infects the eggs of Echmepteryx hageni. Host eggs generally contained a single female and one or more males. The male is the smallest insect so far recorded at 0.139-0.240 mm. The scale bar to the right is 0.1 mm. These bizarre males have no eyes or wings but tarsi that resemble suction cups. Mockford observed males attached to females by thier remarkale tarsi. Describing the male, Mockford says "He has long legs. Although the antennae are unsegmented, they are relatively large, protruding like great ears from the tiny head. He looks like an organism equipped to hunt for something, presumably a mate. Perhaps he is clinging with his suction cups on his sibling female to a new set of host eggs, where he then quests for a nonsibling mate."
Psocid eggs do play host to parasitoid wasps of the family Mymaridae, which lay their eggs in recently deposited host eggs. One remarkable such "fairyfly" was described by E. L. Mockford (Ann. Entomol. Soc. Am. 90(2): 115-120, 1997). Dicopomorpha echmepterygis infects the eggs of Echmepteryx hageni. Host eggs generally contained a single female and one or more males. The male is the smallest insect so far recorded at 0.139-0.240 mm. The scale bar to the right is 0.1 mm. These bizarre males have no eyes or wings but tarsi that resemble suction cups. Mockford observed males attached to females by thier remarkale tarsi. Describing the male, Mockford says "He has long legs. Although the antennae are unsegmented, they are relatively large, protruding like great ears from the tiny head. He looks like an organism equipped to hunt for something, presumably a mate. Perhaps he is clinging with his suction cups on his sibling female to a new set of host eggs, where he then quests for a nonsibling mate."
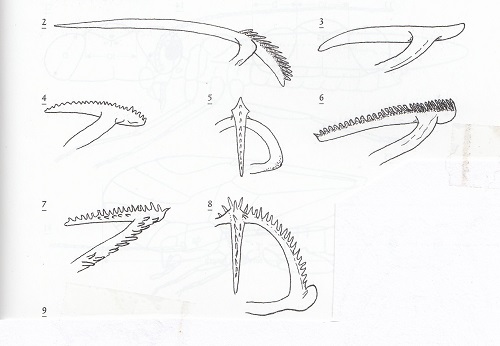 In order to escape from the egg, psocid pronymphs employ a specialized ovirupter or egg burster which is usually armed with numerous teeth. According to Kathryn M. Sommerman who observed this process in Ectopsocopsis cryptomeriae: "The arms of the egg burster stretch across the front of the head with the center puncturing shaft projecting posteriorly towards the clypeus. The heads of several individuals were observed pulsating before the chorions were broken…at the time the chorion splits, the top of the head is pulsating regularly. Fifteen minutes after the chorion was noticably broken the egg burster punctured the pronymphal membrane. The pronymphal exuviae bearing the egg burster always remained partly extruded from the chorion." (Psyche 50 (3-4): 53-66, 1943).
In order to escape from the egg, psocid pronymphs employ a specialized ovirupter or egg burster which is usually armed with numerous teeth. According to Kathryn M. Sommerman who observed this process in Ectopsocopsis cryptomeriae: "The arms of the egg burster stretch across the front of the head with the center puncturing shaft projecting posteriorly towards the clypeus. The heads of several individuals were observed pulsating before the chorions were broken…at the time the chorion splits, the top of the head is pulsating regularly. Fifteen minutes after the chorion was noticably broken the egg burster punctured the pronymphal membrane. The pronymphal exuviae bearing the egg burster always remained partly extruded from the chorion." (Psyche 50 (3-4): 53-66, 1943).
The image to the right shows oviruptors form 2) Stenopsocus, 3) a typical bark-frequenting psocid (Psocidae), 4 & 5) Ectopsocus, 6) Graphopsocus , 7 & 8) Caecilius. These figures are from The Psocoptera of tropical South-east Asia, T. R. New and C. Lienhard, 2007.
Although there have been few published reports of egg guarding in "Psocoptera", numerous observations of females lingering adjacent to egg clusters may indicte that this behavior does occur in some taxa. I have personally observed this behavior in Ectopsocus meridionalis. Teliapsocus conterminus image by Dan Leeder and Polypsocus corruptus image by John Rosenfeld.
